CDC panel: Rare heart inflammation cases don’t warrant overturning vaccine advice

The Pfizer and Moderna COVID-19 vaccines have a “likely association” with a heart condition in some adolescents and young adults, particularly males, members of the nation’s top vaccine advisory panel said Wednesday.
However, such cases have been extremely rare and usually are so mild that the benefits of vaccination far outweigh any potential risks, members of the CDC’s Advisory Committee on Immunization Practices said.
The vaccines have “demonstrated a high degree of safety,” said Dr. Matthew Daley, a panel member and Colorado pediatrician. “The data clearly should give us the confidence that we have systems in place to identify risk.”
Still, to better inform the public, the U.S. Food and Drug Administration plans to provide warning statements for vaccine providers and patients about the potential risk of inflammation of the heart muscle, called myocarditis, or pericarditis, inflammation of tissues around the heart.
Meanwhile, the Centers for Disease Control and Prevention will continue to investigate the reports and monitor cases. Researchers want to understand why most of the reports of myocarditis have been in males under age 30 after the second vaccine dose, what triggers the inflammation and whether there are any lingering health effects.

“Heart function tends to rebound rather quickly, so that’s reassuring,” Dr. Matthew Oster, a pediatric cardiologist at Children’s Healthcare of Atlanta and Emory, said at the advisory panel meeting.
“We must follow patients closely to see that the long-term outcomes are good as well,” he said, then added, “I’m optimistic they’ll do well.”
The committee discussion came after CDC has received more than 800 unsubstantiated reports of myocarditis or pericarditis among those who had received an mRNA vaccine. Such reports were concentrated among those ages 29 and younger, with 484 cases.
That’s a pattern also seen in Georgia, where 13 cases have been reported to the CDC’s Vaccine Adverse Event Reporting System (VAERS), with eight of them among people under age 40. Six of the eight Georgians reportedly hospitalized with the condition were male, according to the reports.
Panel members stressed that such reports are rare. Those 484 reports come after more than 310 million vaccine doses have been administered in the U.S.
Experts also noted that the CDC’s system accepts reports from everyone, regardless of the plausibility that the vaccine was the cause of a perceived problem. The CDC has confirmed that 323 of the 484 reported cases among those under age 30 met the definition of myocarditis or pericarditis, while most of the others are under review.
Myocarditis, on its own, is rare among the general population, studies show. It occurs in about 22 out of 100,000 people every year. However, with adolescents, the rate is much lower. As a result, researchers expressed concerns if cases were occurring at a higher rate than the general rate.
The unknowns
A recent study on the website of the American Academy of Cardiology reported that the myocarditis experienced by patients after COVID-19 vaccination appeared to be generally mild, and that further study is required “to prove a causal relationship and better understand the mechanisms of this process.”
It’s important to study why the condition appears to predominantly affect males, Oster said. Out of every million doses given, 32 males had experienced myocarditis after second doses, compared to fewer than five females, according to the CDC’s vaccine safety datalink.
As to what may be triggering the condition, Oster said there have been only a handful of studies and they have not provided a definitive answer. “It’s hard to say anything about the mechanism,” he said.
Most of the cases came within a few days of the second dose of either mRNA vaccine.
Overall in Georgia, where eight million doses of COVID-19 vaccine have been administered, 13 cases of myocarditis or pericarditis were reported to the CDC's VAERS system as of Tuesday. Eight of the cases were in people under age 40. Among the five cases in older people, only one was considered serious, involving a 69-year-old who suffered pericarditis and was hospitalized for one day.
Five cases were reported among Georgia women. Three were deemed not serious, involving females ages 43, 44 and 52. Two women, ages 29 and 69, were reportedly hospitalized.
Ten of the 13 Georgia cases involved the Pfizer/BioNTech vaccine, the self-reported records showed, while two cases occurred after Moderna vaccination and one the Johnson and Johnson vaccination. CDC data, however, indicated that more cases nationwide may be associated with the Moderna vaccine. But at this point there's not enough data to distinguish whether one vaccine has more of an association with the inflammation issues, panel members said.
The most common presenting symptom appears to be severe chest pain, typically at rest, Dr. William Mahle, chief of cardiology and co-chief of the Children’s Heart Center at Children’s Healthcare of Atlanta, told the Atlanta Journal-Constitution.
Physicians there have experience taking care of adolescents with the condition and have been in discussion with CDC and other hospitals regarding myocarditis after COVID-19 vaccination, he said.
“Prompt recognition of and management of this condition has typically led to a rather quick improvement in symptoms,” he said.
In the southeast coastal region of Georgia, Dr. Stephen Thacker, a pediatric infectious disease specialist at Memorial Health Dwaine & Cynthia Willett Children’s Hospital of Savannah, has been discussing the issue with local pediatricians, though no cases have yet been reported there.
He’s glad to hear that most patients spend only a few days, at most, in the hospital.
“To me, it’s a good story that for those that may be impacted from vaccination and this very rare consequence, the vast majority have a complete recovery,’' Thacker said.
Risks vs. benefits
A portion of the advisory panel’s meeting was devoted to a discussion of the risks of mRNA vaccination for young males vs. the benefits.
Daley and other committee members pointed to the dramatic drop in illness, hospitalizations and deaths since the vaccines were introduced in December.
Late last year, an estimated 250,000 new cases as well as 3,000 deaths were reported each day. As of June, that has dropped to an estimated 13,000 new cases and 300 deaths per day.
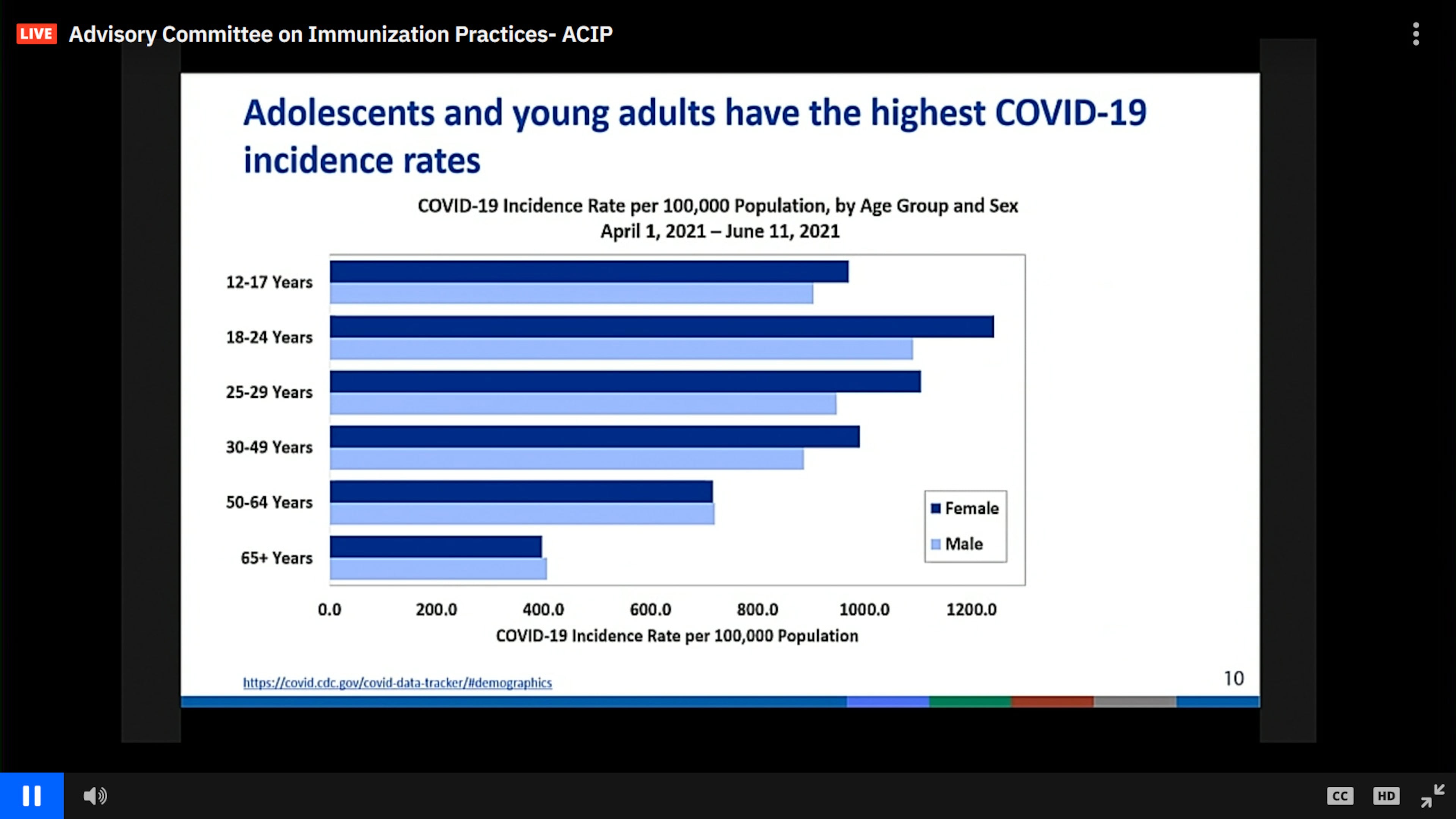
Vaccinations among adolescents and young adults are lagging, though, and committee members noted that while COVID-19 is generally milder in those age groups, adolescents and young adults now have the highest COVID-19 incidence rates.
Second doses of the vaccines are predicted to prevent 12,000 COVID-19 cases among males ages 18-24, and 530 hospitalizations. In contrast, 46 to 56 myocarditis cases are likely to be reported, the panel was told.
To date, Daley said, the vaccines authorized in the U.S. have demonstrated a high degree of safety.
“Nothing in life is absolutely risk-free and we know from decades of experience with other vaccines that rare and potentially serious adverse effects can occur following vaccination,” he said.
However, he said, the nation’s vaccine safety system “is the most rigorous system ever put in placed to monitor any vaccine.” That should give confidence, he said, that if an adverse event arises, the system will identify it.


